One of the most striking features of multicellular organisms is their ability to coordinate information from the various cell types and tissues comprising a single functional organ unit. Unlike animals, plants produce new organs and adjust their growth throughout their life span. In addition, plant cells are held together by a rigid cell wall and do not migrate. These inherently distinct features demand specialized control mechanisms of the growing tissues, to ensure appropriate organ size and shape. How do different cells manage to grow in synchrony, modulate their growth rate and direction? and how this is tuned by a changing environment?
To tackle these questions, our lab studies the role of hormonal signaling, cell identity and mechanical constraints that together dictate developmental output. Our research focuses on roots as a model organ and on the hormone brassinosteroid (BR) signaling pathway as a means to elucidate developmental processes. In particular, the regulation of growth coordination among cells, growth directionality at the cell and organ level and meristematic function.
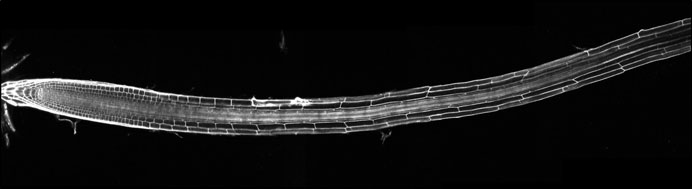
From pleiotropic effects of the hormone to high-resolution and context-specific control of organ growth
Over the past decade, our lab worked towards bridging the gap between the established BR signaling pathway and its less understood interpretation at the cell and organ levels. Using the Arabidopsis root as a model organ, we found that BR signaling regulates the size of the root meristem, cell cycle progression and whole root growth in a cell-specific manner, involving both autonomous and non-autonomous effects (Hacham et al, Development, 2011). We further established that the spatial distribution of BR signaling is an important fine-tuning determinant of root growth. In certain cell types, BR signaling drives cell elongation and cell proliferation, while in others, it restrains these processes (Fridman et al, Genes & Dev, 2014, Vragović et al, PNAS, 2015). This cell-cell coordination likely involves mechanical sensing and mechanical constraints (Fridman et al, Genes & Dev, 2014, and see below) and chemical signals (Vragović et al, PNAS, 2015, and see below). Together, we proposed that BR signaling triggers different developmental programs in different cell types as a means to ensure a coherent organ growth.
Mechanisms for inter-tissue coordination
What is the molecular genetic framework driving the aforementioned differential developmental programs triggered by BR signaling? To tackle this question, we established a unique polyribosome-associated mRNA map of tissue-specific temporal BR responses in various mutant backgrounds, which distinguished between autonomous and non-autonomous responses to the hormone. We revealed a differential effect of the hormone on gene expression in the epidermis versus the innermost tissues (implicated for example, by the magnitude of responsive genes, type of genes, repression vs. activation and gene enrichment along the root zones) (Vragović et al., 2015, PNAS). This mapping provided solid functional genomic evidence of the context-specific impact of the BR hormone.
Assisted by our mapping approach, we revealed a dual effect of BR on auxin and uncovered its critical developmental significance (Ackerman-Lavert et al, Curr Biol, 2021). We demonstrated that BR simultaneously promotes auxin signaling input and represses its output, and revealed that this incoherent effect of BR dictates the root meristematic state. We also showed that this concept also controls meristem regeneration, with Idan Efroni’s lab. Using a broad range of tools we could spatially separate and thus link BR signaling-control of its own production (inner tissues) to BR signaling-control of the meristem function (outer tissues). Hence, the spatiotemporal negative feedback on BR biosynthesis genes is an important developmental determinant of meristem shape and maintenance. Together, we demonstrated a basis for inter-tissue coordination and how a critical ratio between BR and auxin determines the meristematic state.
Cell geometry and biomechanics
In-depth understanding of morphogenesis requires precise single-cell tools that quantify growth parameters in 3D. We quantified the 3D geometry of thousands of individual meristematic cells across different tissue types. We discovered that BR signaling primarily controls cell shape and growth anisotropy, while, surprisingly, maintaining a coherent distribution of cell sizes (i.e. volume) (Fridman et al, Nature Plants, 2021). Using 4D analysis (cellular growth rates in 3D over time) we also demonstrated how cell shape is achieved over time. Thus, our quantitative based-approach defines BR signaling as a controller of directional growth. We then linked geometry at the cellular scale to whole meristem growth, using a computer simulation with Richard Smith’s lab. Based on this model, we proposed that BR signaling controls growth via interaction with tissue-specific mechanical constraints (Fridman et al, Nature Plants, 2021). We also showed together with Sebastian Wolf’s lab, that optimal BR signaling is required for adequate cell wall orientation (Li et al, Development, 2021).
Developmental plasticity in response to changing environment
We uncovered a reciprocal control of mineral nutrients and BR signaling as a means to achieve growth plasticity. Specifically, a molecular interaction and interdependency between BR signaling and the availability of phosphate and iron was found to determine the extent of root growth (Singh et al, 2014; Singh et al, Dev Cell, 2018). Thus, BR regulatory components link nutrient status to root morphology, thereby driving adaptive plant responses.
